What is the USP standard for Gellan Gum?
Gellan Gum Analytical Method
USP29-NF24
Gellan Gum
![]()
![]()
![]() [71010-52-1].
[71010-52-1].
» Gellan Gum is a high molecular weight polysaccharide gum produced by a pure-culture fermentation of a carbohydrate with Pseudomonas elodea, purified by recovery with isopropyl alcohol, and then dried and milled. It is a heteropolysaccharide comprising a tetrasaccharide repeating unit of one rhamnose, one glucuronic acid, and two glucose units. The glucuronic acid is neutralized to mixed potassium, sodium, calcium, and magnesium salts. It may contain acyl (glyceryl and acetyl) groups as the O-glycosidically linked esters. It yields not less than 3.3 percent and not more than 6.8 percent of carbon dioxide, calculated on the dried basis.
Packaging and storage— Preserve in well-closed containers, and store at room temperature.
Identification—
A: Prepare a 1% solution of Gellan gum by hydrating 1g in 99 mL of deionized water. Stir the mixture for about 2 hours, using a motorized stirrer and a propeller-type stirring blade. Draw a small amount of the solution obtained into a wide-bore pipet, and transfer it into a 10% calcium chloride solution. [ NOTE— Reserve the remaining portion of this solution for Identification test B. ] A tough, wormlike gel will form instantly.
B: To the remaining solution prepared for Identification test A, add 0.5 g of sodium chloride, heat the solution to 80![]() , stirring constantly, and hold at 80
, stirring constantly, and hold at 80![]() for 1 minute. Stop heating and stirring the solution, and allow it to cool to room temperature. A firm gel will form.
for 1 minute. Stop heating and stirring the solution, and allow it to cool to room temperature. A firm gel will form.
Microbial limits 61 — It meets the requirements of the tests for absence of Salmonella species and Escherichia coli. The total aerobic microbial count is not more than 1000 cfu per g, and the total combined molds and yeasts count is not more than 100 cfu per g.
Loss on drying 731 — Dry it at 105![]() for 2.5 hours: it loses not more than 15.0%.
for 2.5 hours: it loses not more than 15.0%.
Total ash 561 : not less than 4.0% and not more than 14.0%, calculated on the dried basis.
Arsenic, Method II 211 : not more than 3.0 µg per g.
Lead 251 — Prepare a Test Preparation as directed, using a 2.0-g portion of Gellan Gum. Use 4 mL of Diluted Standard Lead Solution (4 µg of Pb) for the test: the limit is not more than 2.0 µg per g.
Limit of isopropyl alcohol— Proceed as directed under Limit of isopropyl alcohol for Xanthan Gum, using about 5 g of Gellan Gum, accurately weighed, for the Test solution: not more than 750 µg per g is found.
Residual solvents 467 : meets the requirements.
(Official January 1, 2007)
Assay— Proceed with Gellan Gum as directed for Procedure under Alginates Assay 311 , using about 1.2 g of undried Gellan Gum, accurately weighed.
Auxiliary Information— Staff Liaison : Hong Wang, Ph.D. , Senior Scientific Associate
Expert Committee : (EM205) Excipient Monographs 2
USP29–NF24 Page 3338
Pharmacopeial Forum : Volume No. 30(4) Page 1328
Phone Number : 1-301-816-8351
ASSAY ( CO2 Yields)
APPARATUS
The required apparatus (see Figure 2) contains a capillary metering valve, A, followed by a flowmeter, B, to control and monitor the flow of nitrogen through the system. Halogenated vinyl plastic tubing and a rubber fitting, C, are used to connect the flow meter to a sidearm of a reaction flask, D. Flask D is a 250-mL round-bottom, boiling flask, resting in a suitable heating mantle, E. Flask D is provided with a 225-mm Hopkins coil reflux condenser, F. The condenser terminates in a U-shaped trap, G, which contains two 25-g bands of 20-mesh zinc, the bands being bounded and separated by three 3-inch plugs of glass wool. The trap terminates in an adapter, H, that by means of a halogenated vinyl plastic tubing and a twistcock connector, I, connects with a 250-mL gas washing bottle, J. The inlet (bubbling) tube extends almost to the bottom of the gas washing bottle, and it terminates in a fritted disk having a coarse porosity. The size of all glass joints is 24/40, except for the 45/50 joint of the gas washing bottle.
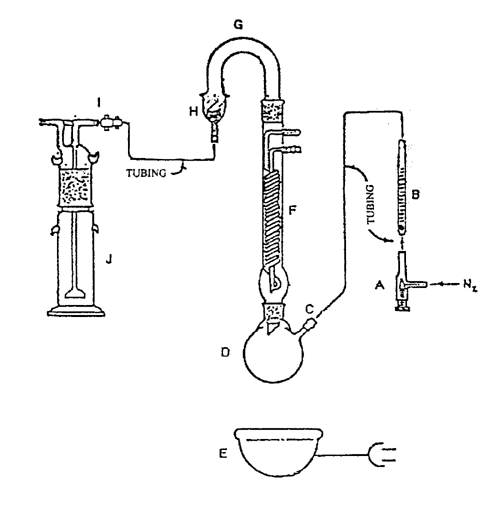
Fig. 2. Apparatus for Assay
SYSTEM SUITABILITY
Using D-glucuronolactone as the standard, proceed as directed for Procedure, but do not perform preboiling steps. The system is suitable if the following criteria are met: (1) a blank determination results in a net titration value C, between 0.02 and 0.06 mEq of 0.1 N hydrochloric acid, calculated as follows:
Ab – Bb
in which Ab is the number of mEq of 0.25 N sodium hydroxide in the 25 mL used, and Bb is the number of mEq of 0.1 N hydrochloric acid used in the blank titration; and (2) the percentage of carbon dioxide, CO2, obtained from the standard is between 24.7% and 25.3%.
PROCEDURE
transfer a specimen of about 1.2g, accurately weighed, into the reaction flask, D, add 50 mL of 0.1 N hydrochloric acid, insert several boiling chips, and connect the flask to the reflux condenser, F, using phosphoric acid as a lubricant. [NOTE—Stopcock grease may be used for the other connections.] Connect the nitrogen line to the sidearm of the flask, and adjust the flow of cooling water to about 2 L per minute.
[NOTE—The following preboiling steps, outlined in this paragraph, are optional and need only be performed when the presence of inorganic carbonates is suspected.] Maintain the flow of nitrogen through the apparatus at 90 to 100 mL per minute. Raise the heating mantle, E, to the flask, heat the specimen to boiling, and boil gently for 2 minutes. Turn the heat off, lower the mantle, E, and allow to cool for about 10 minutes.
Connect the empty gas washing bottle assembly, J, and sweep the system with nitrogen at a rate of 90 to 100 mL per minute for 5 minutes. Reduce the nitrogen flow to 60 to 65 mL per minute, add 10 drops of butyl alcohol, 25.0 mL of 0.25 N sodium hydroxide VS, and 50 mL of distilled water into the bottle, rinsing down the inside of the gas washing bottle, and replace the cap. Detach the rubber fitting, C, from sidearm, and add 46 mL of hydrochloric acid through the sidearm of the boiling flask. Reattach the nitrogen line, raise the heating mantle, and heat the reaction mixture to boiling. After 2 hours of boiling, increase the nitrogen flow to 90 to 100 mL per minute, discontinue the heating, and lower the mantle. Allow to cool for 10 minutes. Disconnect, and disassemble the gas washing bottle. Using a directed stream of distilled water, thoroughly rinse all parts of the bubbling tube and cap, collecting the washings in the gas washing bottle. Use nitrogen to gently force all water out of the bubbling tube. To the bottle immediately add 10 mL of 10% barium chloride solution and a stirring bar. Insert a tight stopper, and stir gently for 1 minute. Allow to stand for at least 5 minutes. Add three drops of phenolphthalein TS, and titrate with 0.1 N hydrochloric acid VS. Perform a blank determination. Calculate the percentage of carbon dioxide, CO2, by the formula:
2200[(A -B) -C] / (1000W) (1 - D)
in which A is the number of mEq of 0.25 N sodium hydroxide in the 25 mL used; B is the number of mEq of 0.1 N hydrochloric acid used for the titration of the sample or the standard; C is the net titration value calculated in the blank determination; W is the weight, in g, of the sample or the standard taken; and D is the percentage expressed as a decimal (1 decimal place), obtained in the test for Loss on drying for the sample or for the standard.