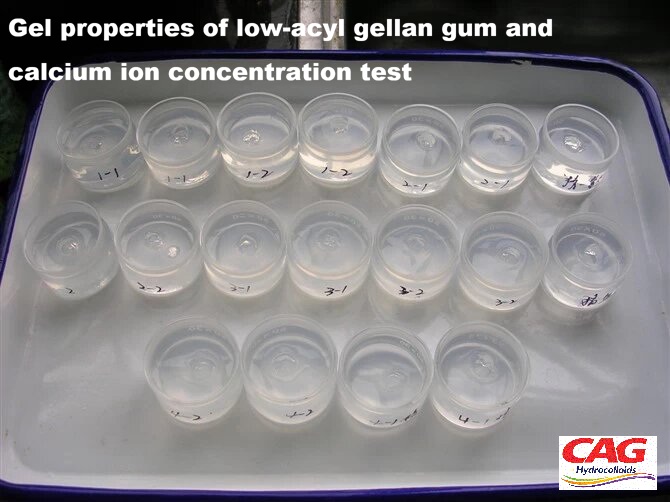
How do ions influence the melting and setting temperatures of gellan gum gels, and what practical application does this enable?
The melting and setting points of gellan gum gels exhibit a strong, predictable dependence on both the type and concentration of cations present. Experimental data indicates that gels formed with calcium ions (Ca²⁺) typically set within a range of 25–40°C, while those formed with sodium ions (Na⁺) set at higher temperatures, between 40–50°C. The ionic concentration has a particularly marked influence on the melting temperature. At lower ion levels, the gels remain thermo-reversible and will re-melt upon heating. However, as the ion concentration increases, the melting point rises significantly. At sufficiently high ion levels, the gels become heat-stable and do not melt below 100°C. This property is critically useful in applications requiring thermal processing, such as in canned foods or bakery products that must withstand high temperatures without losing their gelled structure.