
CAG Company Bacteriological Agar Quality Standard: A Glimpse into Our Benchmarks
Maintaining the highest purity and performance in bacteriological agar is crucial for reliable microbiology results. At CAG, we adhere to stringent, transparent quality standards. Here’s a look at our key testing parameters:
I. Physicochemical Specifications
|
Parameter |
Specification |
|---|---|
|
Moisture |
≤ 5% |
|
Turbidity (NTU) |
≤ 8 |
|
Gel Strength |
900–1000 g/cm² |
|
pH |
6.0–7.5 |
|
Stability at 48°C (4 hours) water bath |
No gel formation or flocculation |
|
Heat-Resistant Bacteria Test (55°C, 72 hours) |
No growth of thermophilic bacteria |
|
Phosphate Precipitation Test |
No precipitation |


Sterilization Operation Heat-Resistant Bacteria Test (55°C, 72 hours) ( Failed Sample )


Stability at 48°C (4 hours) water bath ( Failed Sample ) Phosphate Precipitation Test ( Failed Sample )
II. Microbiological Performance
|
Test Strain |
Acceptance Criteria |
|---|---|
|
Escherichia coliATCC 25922 |
Pass |
|
Staphylococcus aureusATCC 6538 |
Pass |
|
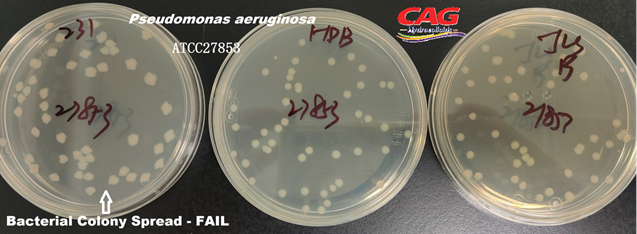
Pseudomonas aeruginosaATCC 27853 |
Colonies must be round, regular, and show no spreading |
|
Bacillus subtilisATCC 6633 |
Pass |
|
Streptococcus pneumoniaeCMCC(B) 31001 |
Pass |
|
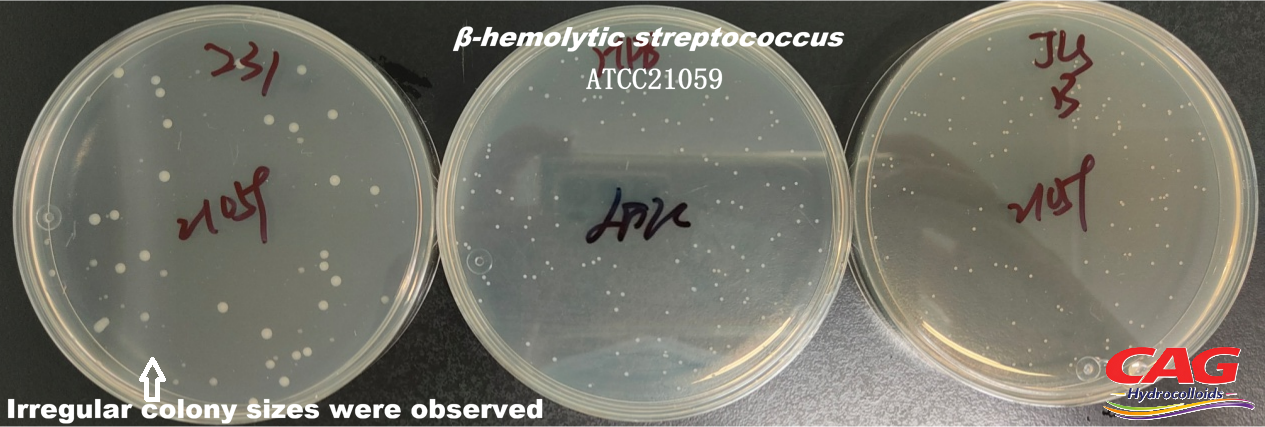
Beta-hemolytic StreptococcusATCC 21059 |
Colonies must be uniform in size |
|
Candida albicansCMCC(F) 98001 |
Pass |
Why This Matters:
Consistency – Tight control of gel strength and pH ensures reproducible media performance.
Clarity – Low turbidity provides better visibility for colony observation.
Stability – No gelling or flocculation at 48°C confirms excellent thermal stability.
Microbiological Purity – Rigorous testing against key ATCC strains ensures reliable growth without interference.
This is how we support labs in achieving accurate, dependable microbial results—batch after batch.